 Kitchens, University of Texas Southwestern Medical Center, Dallas, TX) into CHO-K1 wild-type cells or CHO/CD14 reporter cells ( 16). Stable expression of TLRs was obtained by cotransfection of these epitope-tagged plasmids with pcDNA3 (Invitrogen, San Diego, CA) or pRL/RSV/puro (gift of R. The cDNAs for human TLRs 2 and 4 were the gifts of Carsten Kirschning and Mike Rothe (Tularik, South San Francisco, CA), and were cloned into the vector pFLAG as described ( 8). This clonal line has been cotransfected with CD14 and a NF-κB-dependent reporter plasmid that drives the expression of surface CD25 Ag resulting from LPS-, TNFα-, or IL-1β-induced NF-κB translocation. The engineering of the CD14-expressing Chinese hamster ovary (CHO)-K1 reporter fibroblast cell line CHO/, also known as clone 3E10, has been previously described in detail ( 15). Materials and MethodsĪll cell lines were grown as adherent monolayers at 37☌ in a 5% saturated CO 2 atmosphere, and were passaged at least twice weekly to maintain logarithmic growth. The use of common receptor systems suggests that the often observed clinical parallels between Gram-positive and Gram-negative bacterial infection result from the activation of similar signal transduction systems. 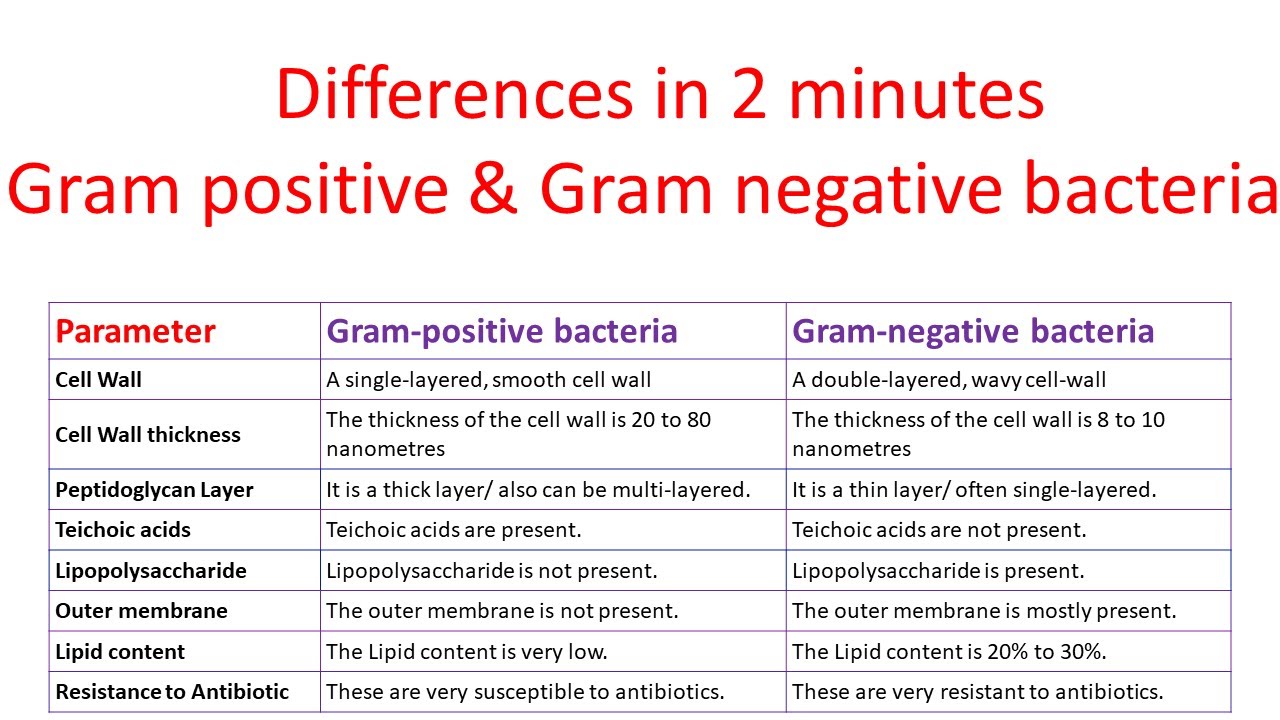
Furthermore, the recognition of these bacteria, at least in part, occurs via the PGN skeleton. In contrast, TLR4 appears to be excluded as a component of a receptor system involved in the recognition of these types of bacteria. We report here that the coexpression of CD14 and human TLR2 resulted in the recognition of two distinct and clinically important genuses of Gram-positive bacteria. Given the similarity in responses to exposure to Gram-positive bacteria and Gram-negative bacteria, and the common dependence on many of their cell wall products upon CD14, we hypothesized that the downstream elements of the signal transduction system might consist of common genetic elements. 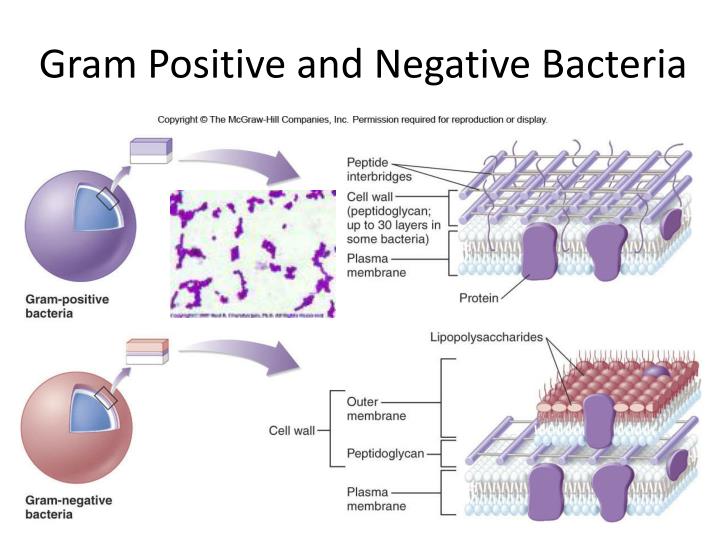
Both peptidoglycan (PGN) and lipoteichoic acid have been demonstrated to activate macrophages in a CD14-dependent manner ( 12, 13). Like Gram-negative bacteria, major components of the Gram-positive bacterial cell wall employ CD14 for immune recognition. This suggests the hypothesis that the true role of TLR2 is the recognition of other bacterial ligands that in some way are similar to LPS. Indeed, the LPS hyporesponder phenotype of C3H/HeJ mice is so profound that, despite the LPS signaling capability of TLR2, it seems likely that TLR4 is the major mammalian LPS signal transducer. TLR4 cloned from the C3H/HeJ mouse proved to harbor a point mutation that rendered it nonfunctional ( 9, 10, 11), consistent with the concept the mutant TLR4 might function as a dominant-negative mutation accounting for LPS hyporesponsiveness in the C3H/HeJ mouse. Lps mapped to the same region as TLR4 ( 9, 10). Additional evidence that TLRs function as LPS signal transducers comes from positional cloning of Lps, the genetic locus for LPS sensitivity that is abnormal in C3H/HeJ mice. This activity of TLR2 was potentiated by CD14, the LPS-binding receptor. ( 8) recently demonstrated that a human homologue of Toll, known as Toll-like receptor 2 (TLR2), 3 apparently functions as an LPS signal transducer when transfected into LPS nonresponder cell lines. For example, the Toll homologue known as 18-wheeler is responsible for responses to Gram-negative bacteria ( 5), whereas Toll regulates antifungal responses ( 6). The family of Toll proteins appears to be responsible for specific immune recognition in Drosophila melanogaster. These data suggest that the similarity of clinical response to invasive infection by Gram-positive and Gram-negative bacteria is due to bacterial recognition via similar TLRs.Īlthough the exact mechanism of immune activation by Gram-positive bacteria remains unknown, recent studies of immune activation by bacterial LPS provide a clue. 
Soluble peptidoglycan substituted for whole organisms. To determine which components of Gram-positive cell walls activate Toll proteins, we tested a soluble preparation of peptidoglycan prepared from S. CD14 coexpression synergistically enhanced TLR2-mediated activation. Heterologous expression of human TLR2, but not TLR4, in fibroblasts conferred responsiveness to Staphylococcus aureus and Streptococcus pneumoniae as evidenced by inducible translocation of NF-κB. We hypothesized that Gram-positive bacteria might also be recognized by TLRs. The LPS component of the Gram-negative bacterial cell wall appears to activate cells via CD14 and Toll-like receptor (TLR) 2 and TLR4. The basis for the earliest steps in innate immune response to Gram-positive bacterial infection is poorly understood. Invasive infection with Gram-positive and Gram-negative bacteria often results in septic shock and death. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
